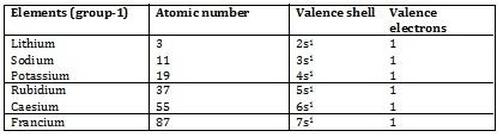
The alkaline earth metals (group II) are always assigned an oxidation number of +2.The alkali metals (group I) always have an oxidation number of +1.Fluorine in compounds is always assigned an oxidation number of -1. 
The oxidation number of a monatomic ion equals the charge of the ion.The oxidation number of a free element is always 0.You can find examples of usage on the Divide the redox reaction into two half-reactions page. Since the electrons between two carbon atoms are evenly spread, the R group does not change the oxidation number of the carbon atom it's attached to. Unlike radicals in organic molecules, R cannot be hydrogen. Organic compounds can be written in such a way that anything that doesn't change before the first C-C bond is replaced with the abbreviation R (Figure 1c). When dealing with organic compounds and formulas with multiple atoms of the same element, it's easier to work with molecular formulas and average oxidation numbers (Figure 1d). Notice that changing the CH 3 group with R does not change the oxidation number of the central atom. R is an abbreviation for any group in which a carbon atom is attached to the rest of the molecule by a C-C bond. Different ways of displaying oxidation numbers of ethanol and acetic acid. Taken as a whole, the atom at the bottom of a u-bond is saying "I don't like it, but if you are that desperate for a pair of electrons, and if you promise to stay very close by, I'll let you latch onto a pair of electrons from my already-completed octet.Figure 1. Notationally, this is reflected by the fact that both of the line ends of the U are next to that atom. The beggar atom at the top of the U gets to use both of the electrons for free, which in turn means that two of its valence-bond needs are met. This lack of bond completion is represented by the absence of any line ends on that side of the u-bond. That atom gets nothing out of the deal, so none of its bonding needs are changed or satisfied. However, in a u-bond, it is the atom at the bottom of the U that donates both electrons in the pair. 
As with the straight-line covalent bond, the line that forms the u-bond represents a single pair of electrons. The main advantage of u-bond notation is that it lets you prototype and assess non-standard bonding relationships while using only standard atomic valences. I invented it out of frustration in high school back in the 1970s (yes I'm that old), but never did anything with it at the time. The second u-bond notation is the novel one. The upper straight-line equivalence is trivial since the resulting line is identical in appearance and meaning to the standard covalent bond of organic chemistry. The donation notation shows how an octet perspective can still be applied to such cases, though it is never a good idea to rely on first-order approximation models for such extreme cases.įinally, if you are tired of dots and arrows and yearn for something closer to standard valence bond notation, these two equivalences come in handy: Sulfur hexafluoride, $\ce$ be modeled as if the sulfur is a metal that gives up all of its electrons to the hyper-aggressive fluorine atoms (option a), or as a case where the octet rule gives way to a weaker but still workable 12-electron rule (option b)? There is some controversy even today about how such cases should be handled. Manishearth's excellent answer explores this issue in much greater detail than I do here. However, the same bonding mechanisms that enable the formation of greater-than-8 valence shells also enable alternative structural interpretations of such shells, depending mostly on whether such bonds are interpreted as ionic or covalent. If there are sufficiently strong compensating energy factors, even atoms that strongly prefer octets can form stable compounds with more (or less) than the 8 valence shell electrons. While having an octet of valence electrons creates an exceptionally deep energy minimum for most atoms, it is only a minimum, not a fundamental requirement.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
